Newsletter Mar2026. How the Gut Influences Hormones and Metabolism
Along the human digestive tract, there is a complex community of microorganisms known as the microbiome. These microbes include bacteria, viruses, fungi, and other microorganisms. Many of these microbes are beneficial and perform essential functions that support overall health, including digestion of food, immune system regulation, hormone metabolism, and the production of vitamins and metabolic compounds (1,2). The microbiome also interacts with the nervous system through the gut-brain axis, a communication network that can influence mood, stress response, sleep, and cognitive function (3).
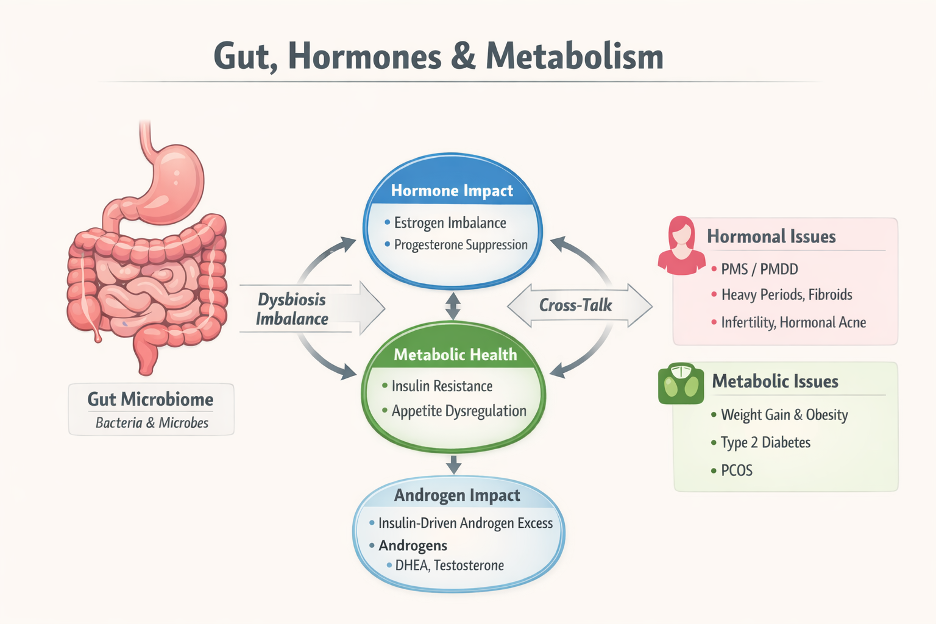
The microbiome functions best when there is a wide diversity of beneficial microbes. However, a diet high in processed, packaged, and sugar-rich foods, along with chronic stress, poor sleep, environmental toxins, and certain medications, can disrupt this healthy balance. This disruption is often referred to as dysbiosis (1,2).
One way to visualize this is to imagine a healthy microbiome as a lush, green lawn. When the microbial ecosystem is balanced, beneficial organisms dominate and maintain stability. When dysbiosis occurs, patches of “bare soil” appear where opportunistic microbes, like weeds in a lawn, can grow and take over. This imbalance may contribute to a variety of symptoms, including fatigue, mood disorders, weight gain, brain fog, digestive concerns, skin rashes or hives, inflammation, low libido, and other signs of hormone imbalance (1,2,3,4).
Certain gut bacteria also participate in the metabolism and regulation of sex hormones such as estrogen and progesterone. These microbes are collectively known as the estrobolome, a group of bacteria that produce enzymes such as beta-glucuronidase, which help regulate the breakdown and elimination of estrogen (4,5). Beta-glucuronidase is a marker that can be evaluated through specialized laboratory testing.
When microbial balance is disrupted, elevated beta-glucuronidase activity can lead to the reactivation and recirculation of estrogen in the body rather than proper elimination through the stool. This process may contribute to a pattern of relative estrogen excess (4,5).
Excess estrogen from a disrupted estrobolome can contribute to conditions such as (3,4,5,6):
· Premenstrual syndrome (PMS)
· Premenstrual dysphoric disorder (PMDD)
· Heavy or painful menstrual cycles
· Uterine fibroids
· Endometriosis
· Infertility or implantation challenges
· Hormonal acne
· Increased symptom severity during perimenopause
Additionally, dysbiosis can promote the production of inflammatory cytokines and activate chronic stress signaling pathways that may suppress progesterone production (3). This process occurs through effects on the hypothalamic-pituitary-ovarian (HPO) axis, sometimes referred to as the gonadal-brain axis, as well as through immune system activation. Together, these processes can further contribute to hormone imbalance and cycle-related symptoms (3,4,5,6).
Another way the microbiome influences hormone balance is through metabolic and endocrine signaling, including insulin sensitivity, appetite hormones, and androgen production (7,8). Androgen hormones, such as DHEA and testosterone, are precursors that can be converted into estrogens within the body. Disruptions in microbial balance affecting metabolic and androgen pathways have been associated with several health conditions (8,9,10), including:
· Polycystic Ovary Syndrome (PCOS)
· Insulin Resistance
· Metabolic Syndrome (Abdominal obesity, high blood sugar, high blood pression, high triglycerides, and low-serum high density lipoprotein (low-HDL aka good cholesterol)
· Weight gain or obesity
· Type 2 Diabetes
When androgen levels become elevated in women, symptoms such as acne, irregular cycles, or excess hair growth may occur. These patterns can reflect disruptions in metabolic and hormonal signaling pathways that are influenced in part by the microbiome.
One important mechanism involves the microbial fermentation of dietary fiber, which produces short-chain fatty acids that help regulate glucose metabolism, inflammation, and insulin sensitivity (7,8). Dysbiosis may also contribute to increased intestinal permeability and systemic inflammation, both of which can impair metabolic signaling and promote metabolic dysfunction (9,10). Over time, chronic insulin resistance may stimulate increased ovarian androgen production, contributing to hyperandrogenism, a hallmark feature commonly seen in PCOS (8,9,10). Together, these mechanisms illustrate how microbiome alterations can influence both metabolic health and reproductive hormone balance.
How do we evaluate and support this?
In our office, we take a holistic approach to evaluating gut health, hormone balance, and metabolic function. One of my preferred tools is an in-depth stool analysis, such as the GI-MAP, which differs from a conventional stool test by evaluating a wider range of commensal and opportunistic bacteria as well as markers of intestinal health. These markers may include beta-glucuronidase (related to estrogen metabolism), calprotectin (a marker of inflammation), immune function indicators, and markers related to intestinal permeability.
We may also recommend advanced urine testing, such as an Organic Acids Test or a dried hormone analysis, which can provide insight into endocrine and metabolic pathways, toxin exposure, and nutrient status. In addition, comprehensive blood testing including fasting insulin and glucose, lipid panels, vitamin D levels, and hormone markers can provide valuable information about metabolic and hormonal health.
Depending on each individual’s needs and underlying conditions, we may recommend targeted nutrition strategies, therapeutic herbs, nutrient support, and lifestyle interventions designed to restore microbial balance and support optimal physiological function.
References
Lynch SV, Pedersen O. The human intestinal microbiome in health and disease. New England Journal of Medicine. 2016;375(24):2369-2379.
Valdes AM, Walter J, Segal E, Spector TD. Role of the gut microbiota in nutrition and health. BMJ. 2018;361:k2179.
Cryan JF, O’Riordan KJ, Cowan CSM, et al. The microbiota-gut-brain axis. Physiological Reviews. 2019;99(4):1877-2013.
Plottel CS, Blaser MJ. Microbiome and malignancy: The estrobolome and estrogen metabolism. Cell Host & Microbe. 2011;10(4):324-335.
Baker JM, Al-Nakkash L, Herbst-Kralovetz MM. Estrogen–gut microbiome axis: Physiological and clinical implications. Maturitas. 2017;103:45-53.
Flores R, Shi J, Fuhrman B, et al. Fecal microbial determinants of fecal and systemic estrogens and estrogen metabolites. Journal of Translational Medicine. 2012;10:253.
Tremaroli V, Bäckhed F. Functional interactions between the gut microbiota and host metabolism. Nature. 2012;489(7415):242–249.
Torres PJ, Siakowska M, Banaszewska B, et al. Gut microbial diversity in women with polycystic ovary syndrome correlates with hyperandrogenism. Journal of Clinical Endocrinology & Metabolism. 2018;103(4):1502–1511.
Canfora EE, Meex RCR, Venema K, Blaak EE. Gut microbial metabolites in obesity, NAFLD and type 2 diabetes. Nature Reviews Endocrinology. 2019;15(5):261–273.
Cani PD, Jordan BF. Gut microbiota-mediated inflammation in obesity: A link with gastrointestinal cancer. Nature Reviews Gastroenterology & Hepatology. 2018;15(11):671–682.
Disclaimer - This document is for informational purposes only and is not intended to provide medical advice, diagnosis, or treatment. Always consult with a qualified healthcare professional before making any changes to your health regimen.